|
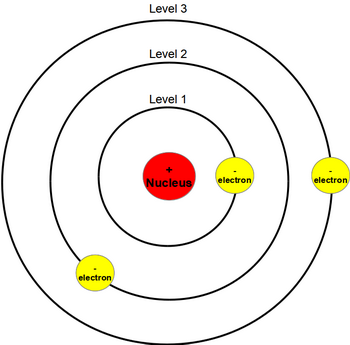
The quantity in parentheses is a unit of energy known as a Rydberg, which \(\approx 13.6eV\).īohr reasoned that the hydrogen atom doesn't radiate away all of its energy, because the lowest energy level (which we would now call the ground state) corresponds to one wavelength fitting in the orbit, so \(n=1\) is the lowest it can go. 
, each of which is associated with an electron binding energy, E. , can be solved to yield a series of wave function. "Sommerfeld formula and Dirac's theory" (PDF). Erwin Schrödinger proposed the quantum mechanical model of the atom, which treats electrons as matter waves.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
